ตำรายาของประเทศไทย
Thai Pharmacopoeia
สำนักยาและวัตถุเสพติด กรมวิทยาศาสตร์การแพทย์ กระทรวงสาธารณสุข
Bureau of Drug and Narcotic, Department of Medical Sciences, Ministry of Public HealthSalol
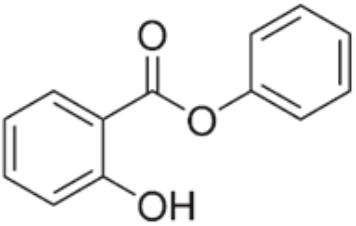
C13H10O3 214.22 118-55-8
Benzoic acid, 2-hydroxy, phenyl ester.
Category Intestinal antiseptic; antibacterial.
Phenyl Salicylate contains not less than 99.0 per cent and not more than 101.0 per cent of C13H10O3.
Description Colourless crystals or a white microcrystalline powder; odour, faint and aromatic.
Solubility Practically insoluble in water; freely soluble in ethanol; slightly soluble in glycerol.
Contra-indication It is contra-indicated in patients with history of hypersensitivity reactions to phenyl salicylate.
Packaging and storage Phenyl Salicylate shall be kept in tightly closed containers, protected from light, and stored at a temperature not exceeding 30º.
Identification
Melting range 42º to 43.5º (Appendix 4.3).
Acidity To a 5 per cent w/v solution in carbon dioxide-free water add phenol red TS. The solution is colourless and not more than 0.20 mL of 0.1 M sodium hydroxide is required to change the colour of the solution.
Chloride Not more than 360 ppm (Appendix 5.2). A 500-mg sample shows no more chloride than that corresponds to 0.25 mL of 0.020 M hydrochloric acid VS.
Sulfate Not more than 0.12 per cent w/w (Appendix 5.2). A 500-mg sample shows no more sulfate than that corresponds to 0.62 mL of 0.010 M sulfuric acid VS.
Limit of phenol and free salicylic acid Not more than 0.10 per cent w/w for phenol and for free salicylic acid. Carry out the test as described in the “Liquid Chromatography” (Appendix 3.5).
Diluent Prepare a mixture of equal volumes of acetonitrile and a 0.03 per cent v/v solution of phosphoric acid.
Standard solution Dissolve an accurately weighed quantity of Phenol RS and Salicylic Acid RS in Diluent to obtain a solution having a known concentration of 2 µg per mL of phenol and salicylic acid.
Test solution Transfer about 200 mg of Phenyl Salicylate, accurately weighed, to a 100-mL volumetric flask, add sufficient Diluent, sonicate for 2 minutes, and dilute with Diluent to volume and mix.
Mobile phase
Mobile phase A Prepare a 0.03 per cent v/v solution of phosphoric acid.
Mobile phase B Acetonitrile.
The step gradient of mobile phases is as follows.
Time (Minutes) | Mobile Phase A (Per Cent V/V) | Mobile phase B (Per Cent V/V) |
0.00 | 80 | 20 |
4.00 | 80 | 20 |
4.10 | 20 | 80 |
6.50 | 20 | 80 |
6.51 | 80 | 20 |
9.00 | 80 | 20 |
Chromatographic System
Detector Ultraviolet light (275 nm for Phenol and 235 nm for Salicylic acid).
Column A stainless steel column (75 mm × 4.6 mm), packed with octadecylsilane chemically bonded to porous silica or ceramic microparticles (3.5 µm).
Temperature
Column About 35º.
Flow rate 2.0 mL per minute.
System suitability
Sample Standard solution.
Suitability requirements
Symmetry factor Not more than 2.0 for the phenol and salicylic acid peaks.
Resolution Not less than 2.0 between the phenol and salicylic acid peaks.
Relative standard deviation Not more than 2.0 per cent for the phenol and salicylic acid peaks.
COLUMN EFFICIENCY Not less than 2,000 theoretical plates for the phenol and salicylic acid peaks.
Procedure Separately inject equal volumes (about 20 µL) of Standard solution and Test solution into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Identify the peaks due to phenol and salicylic acid in the test solution, using the retention times of phenol and salicylic acid in the standard solution.
Calculation Calculate the contents of C6H6O and C7H6O3 in the portion of the Phenyl Salicylate taken, using the declared contents of C6H6O in Phenol RS and C7H6O3 in Salicylic Acid RS.
Assay Carry out the determination as described in the “Liquid Chromatography” (Appendix 3.5).
Diluent Acetonitrile and water (75:25).
Standard preparation Dissolve an accurately weighed quantity of Phenyl Salicylate RS, in Diluent to obtain a solution having a known concentration of 600 µg per mL.
Assay preparation Transfer about 60 mg of Phenyl Salicylate, accurately weighed, to a 100-mL volumetric flask, dissolve in and dilute with Diluent to volume, and mix.
Mobile phase Acetonitrile and water (50:50).
Chromatographic system
Detector Ultraviolet light (275 nm).
Column A stainless steel column (150 mm × 4.6 mm), packed with octadecylsilane chemically bonded to porous silica or ceramic microparticles (5 µm).
Temperature
Column About 35º.
Flow rate 2.0 mL per minute.
System suitability
Sample Standard preparation.
Suitability requirements
Symmetry factor Not more than 2.0.
Relative standard deviation Not more than 2.0 per cent.
COLUMN EFFICIENCY Not less than 2,000 theoretical plates.
Procedure Separately inject equal volumes (about 10 L) of Standard preparation and Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks.
Calculation Calculate the content of C13H10O3 in the portion of the Phenyl Salicylate taken, using the declared content of C13H10O3 in Phenyl Salicylate RS.